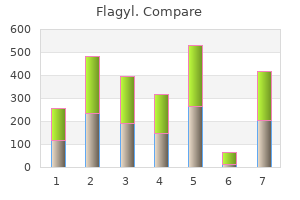
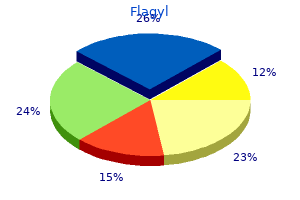
Flagyl"Discount 200mg flagyl visa, bacteria 2014". By: J. Renwik, M.B.A., M.D. Vice Chair, University of Houston Insights into how the body deals with high fat and high sugar diets are emerging from a variety of studies antibiotic for uti gram negative rods flagyl 250mg fast delivery, and evidence points clearly to the benefits of exercise and dietary restriction. Endurance training (such as distance running) and resistance training (with weights) are both beneficial. Endurance training increases the mass of slow-twitch muscle fibers, resistance training builds fast-twitch muscle fibers, and both types of exercise reduce body fat, but in quite different ways. Effects of endurance training include increased expression of (1) peroxisome proliferator-activated receptor d, a transcription factor that builds slow-twitch muscle fiber, and (2) insulin receptors and glucose transporters. Fast-twitch muscles are adapted for short bursts of energy, which can be supplied by glycolysis and thus are termed glycolytic. Effects of resistance training include activation of metabolic processes by the serine/threonine kinase Akt1. Induction of the Akt1 pathway results in growth of fast-twitch skeletal muscle fibers and subsequent effects on several other organs. These include increased fat uptake and oxidation by the liver and heart, reduction of adipose (fat cell) mass, and reduced blood glucose and insulin levels. Weight training Slow-twitch muscle growth Fast-twitch muscle growth Muscle: Increased glucose uptake and glycolysis Local effects in muscle: Increased fatty acid oxidation Increased expression of insulin receptors and glucose transporters Circulating factors Heart: Increased fat uptake and oxidation Adipose tissue: Reduced cell size Circulation: Reduced glucose, insulin, and leptin levels Liver: Increased fat uptake and oxidation yields two molecules of acetyl-CoA in the final step. Alternatively, acetylCoA can also be used as a substrate in amino acid biosynthesis (see Chapter 25). As noted in Chapter 22, however, acetyl-CoA cannot be used as a substrate for gluconeogenesis. The large energy yield from fatty acid oxidation is a reflection of the highly reduced state of the carbon in fatty acids. Sugars, in which the carbon is already partially oxidized, produce less energy, carbon for carbon, than do fatty acids. The breakdown of fatty acids is regulated by a variety of metabolites and hormones. Details of this regulation are described in Chapter 24, following a discussion of fatty acid synthesis. Although some birds migrate over landmasses and eat frequently, other species fly long distances without stopping to eat. The ruby-throated hummingbird, which winters in Central America and nests in eastern North America, often flies nonstop across the Gulf of Mexico. These and similar birds accomplish these prodigious feats by storing large amounts of fatty acids (as triacylglycerols) in the days before their migratory flights. The percentage of dry-weight body fat in these birds may be as high as 70% when migration begins (compared with values of 30% and less for nonmigratory birds). For certain animals-including desert animals (such as gerbils) and killer whales (which do not drink seawater)-the oxidation of fatty acids can be a significant source of dietary water. Metabolism of fatty acids from this store provides needed water (as well as metabolic energy) during periods when drinking water is not available. It might well be said that "the ship of the desert" sails on its own metabolic water! George Holton/Getty Images Eric and David Hosking/Corbis 812 Chapter 23 Fatty Acid Catabolism O 23. Humans and animals whose diets include these food sources metabolize odd-carbon fatty acids via the b-oxidation pathway. The final product of b-oxidation in this case is the three-carbon propionyl-CoA instead of acetyl-CoA. The pKa for the proton that must dissociate to initiate this reaction is approximately 21! Transfer of a hydrogen atom from the substrate to the deoxyadenosyl group produces a methylmalonyl-CoA radical, which then can undergo a classic B12-catalyzed rearrangement to yield a succinyl-CoA radical. Hydrogen transfer from the deoxyadenosyl group yields succinyl-CoA and regenerates the B12 coenzyme (see problem 16 at the end of the chapter). Papain Cholinesterase Optimum pH of Some Enzymes Relative activity Pepsin Trypsin Enzyme Pepsin Catalase Trypsin Fumarase Ribonuclease Arginase Optimum pH 1 antibiotics made simple purchase 500 mg flagyl mastercard. Several effects are operating here: (1) the characteristic increase in reaction rate with temperature; (2) a temperature-dependent equilibrium between active enzyme (Eactive) and the catalytically inactive but not denatured state of the enzyme (Einact); and (3) thermal denaturation of protein structure at higher temperatures. Danson fits well with experimentally observed responses of enzyme activity versus temperature, Keq kinact Eactive 34 Einact 8888n X where X designates the irreversibly inactivated form of the enzyme and k inact is the first-order rate constant for thermal denaturation. Some enzymes, those catalyzing reactions having very high activation energies, show proportionally greater Q10 values. The increasing rate with increasing temperature is ultimately offset by the temperature-dependent equilibrium between active and inactive states of the native enzyme, and, ultimately, the denaturation of protein structure at elevated temperatures. The fit of this model with the experimental data supports the existence of Einact. If the velocity of an enzymatic reaction is decreased or inhibited by some agent, the kinetics of the reaction obviously have been perturbed. Systematic perturbations are a basic tool of experimental scientists; much can be learned about the normal workings of any system by inducing changes in it and then observing the effects of the change. The study of enzyme inhibition has contributed significantly to our understanding of enzymes. Reversible inhibitors interact with the enzyme through noncovalent association/dissociation reactions. In contrast, irreversible inhibitors usually form covalent bonds with side chains or prosthetic groups in the enzyme. That is, the consequence of irreversible inhibition is a decrease in the concentration of active enzyme. The kinetics observed are consistent with this interpretation, as we shall see later. Competitive inhibitors are characterized by the fact that the substrate and inhibitor compete for the same binding site on the enzyme, the Copyright 2017 Cengage Learning. Thus, increasing the concentration of S favors the likelihood of S binding to the enzyme instead of the inhibitor, I. The effects of the other major types, noncompetitive and uncompetitive inhibition, cannot be overcome by increasing [S]. This condition leads us to anticipate that S and I must share a high degree of structural similarity because they bind at the same site on the enzyme. The diagnostic criterion for competitive inhibition is that Vmax is unaffected by I; that is, all lines share a common y-intercept. Note that when [S] is infinitely large (1/[S] < 0), Vmax is the same, whether I is present or not. Obviously, then, the inhibitor is not binding to the same site as S, and the inhibition cannot be overcome by raising [S]. Pure Noncompetitive Inhibition In this situation, the binding of I by E has no effect on the binding of S by E. That is, S and I bind at different sites on E, and binding of I does not affect binding of S. Mixed Noncompetitive Inhibition In this situation, the binding of I by E influences the binding of S by E. Either the binding sites for I and S are near one another or conformational changes in E caused by I affect S binding. A reasonable explanation is that the inhibitor is binding at a site distinct from the active site yet is influencing the binding of S at the active site. Uncompetitive Inhibition Completing the set of inhibitory possibilities is uncompetitive inhibition. A clinically important example is the action of lithium in alleviating manic depression; Li1 ions are uncompetitive inhibitors of myo-inositol monophosphatase. Buy flagyl overnight delivery. MWV Episode 54 - TWiM #16: ICAAC Live.
To add leucine to Lys-Ala to form a linear tripeptide bacteria 30 000 discount 250 mg flagyl with mastercard, precautions must be taken to prevent the incoming Leu a-carboxyl group from reacting inappropriately with the Lys e-amino group instead of the Lys a-amino group. Unlike any amino acid side chain, the phosphate group (or more appropriately, the phosphoryl group) bears two equivalents of negative charge at physiological pH. Furthermore, replacing an H atom on an S, T, or Y side chain with a phosphoryl group introduces a very bulky substituent into the protein structure where none existed before. The Ramachandran plot reveals allowable values of f and c for a-helix and b-sheet formation. The plots consider steric hindrance and will be somewhat specific for individual amino acids. For example, peptide bonds containing glycine can adopt a much wider range of f and c angles than can peptide bonds containing tryptophan. Each of the 60-kD subunits in turn is a heterodimer of two peptides, one of 34 kD and one of 26 kD, joined by at least one disulfide bond. Glycines are essential components of tight turns (b-turns) and, thus, are often essential for maintenance of protein structure. The ability of poly-Glu to form a-helices requires that the glutamate carboxyls be protonated. Deprotonation produces a polyanionic peptide that is not amenable to helix formation. This would allow the two helices of a coiled-coil structure to lie side by side with no twist about the coiled-coil axis. Such a structure would probably not be as stable as the twisted structure of coiled coils formed from a-helices with 3. A sequence containing a phosphorylatable tyrosine and a proline-rich sequence are two examples. Linear motifs are different from domains in the sense that the linear sequence rather than a folded domain is the recognizable unit. The work of Dorothee Kern has provided evidence that conformational transitions in proteins may be assisted by transient interactions resulting from internal protein motions. Consult the reference provided for more information on this interesting phenomenon. He became a professor of physics and chemistry at Howard University in 1941 and spent 27 years on that faculty. In 1948, he took a sabbatical leave in the laboratory of Linus Pauling at the California Institute of Technology. Pauling asked him to do a mathematical analysis of all helical structures for peptide chains that would fit the structural constraints determined previously by Pauling and Robert Corey. The third sequence would place hydrophobic residues on both sides of a b-strand and thus could be found in a parallel b-sheet. The second sequence would place hydrophobic residues on just one side of a b-strand and thus could be found in an antiparallel b-sheet. The first sequence consists of seven-residue repeats, with first and fourth residues hydrophobic, and thus could be part of a coiled coil structure. For the folded state (F) to unfolded state (U) reaction, F 34 U the equilibrium constant is given by Keq 5 u/f 5 u/(1 2 u), where u and f are the unfolded and folded fractions; u 1 f 5 1. The systematic name for trehalose is a-d-glucopyranosyl(1 8n 1)-a-d-glucopyranoside. Methylation of such a sample should yield 1,2,3,6-tetramethylglucose for the glucose residues on the reducing ends of the sample. There are several target sites for trypsin and chymotrypsin in the extracellular sequence of glycophorin, and it would be reasonable to expect that access to these sites by trypsin and Copyright 2017 Cengage Learning. Abbreviated Answers to Problems A-7 chymotrypsin would be restricted by the presence of oligosaccharides in the extracellular domain of glycophorin. Energy yield upon combustion (whether by metabolic pathways or other reactions) depends on the oxidation level. Carbohydrate and protein are at approximately the same oxidation level, and both of these are significantly less than that of fat.
A second virus 68 symptoms 2014 purchase flagyl with a visa, more difficult, approach attempts the completely new design of a protein with the desired structure and activity. Often, this approach relies on in silico methods, where the spatial and reactive properties of its putative active site are modeled, refined, and optimized via computer. Although this approach has fewer limitations in terms of size and shape of substrates, it brings other complications, such as the design of a proper protein scaffold for the active site, protein folding, and protein stability, to say nothing of the difficulties of going from the computer model (in silico) to a real enzyme in a cellular environment (in vivo). Nevertheless, computer design of enzymes capable of catalyzing a reaction at rates up to 6 3 108 times the uncatalyzed rate has been achieved. The Rosetta protein modeling suite is popular software for such in silico enzyme design. New enzyme functions have appeared time and time again, as mutation and selection according to Darwinian principles operate on existing enzymes. Some enzyme designers have coupled natural evolutionary processes with rational design using in vitro mutagenesis. Expression of mutated versions of the gene encoding the enzyme in bacteria, followed by rounds of selection for bacteria producing an enzyme with even better catalytic properties, takes advantage of naturally occurring processes to drive further mutation and selection for an optimal enzyme. This dual approach is whimsically referred to as semirational design because it relies on the rational substitution of certain amino acids with new ones in the active site, followed by directed evolution (selection for bacteria expressing more efficient versions of the enzyme). Degradation of chlorinated epoxides is a major problem in the removal of toxic pollutants from water resources. Genetic engineering of an epoxide hydrolase to create an enzyme capable of using cis-1,2-dichloroepoxyethane as a substrate and catalyzing step 2 is a practical example of de novo enzyme design. Summary 471 hydrolase at three active-site residues (F108, I219, and C248) and selection in bacteria for enhanced chlorinated epoxide hydrolase activity yielded an F108L, I219L, C248I mutant enzyme that catalyzed the conversion of cis-dichloroepoxyethane to Cl2 ions and glyoxal with a dramatically increased Vmax/K m ratio. Enzymes provide kinetic control over thermodynamic potentiality: Reactions occur in a timeframe suitable to the metabolic requirements of cells. Enzymes can be characterized in terms of three prominent features: catalytic power, specificity, and regulation. The site on the enzyme where substrate binds and catalysis occurs is called the active site. Regulation of enzyme activity is essential to the integration and regulation of metabolism. Enzyme kinetics can determine the maximum reaction velocity that the enzyme can attain, its binding affinities for substrates and inhibitors, and the mechanism by which it accomplishes its catalysis. The kinetics of simple chemical reactions provides a foundation for exploring enzyme kinetics. Enzymes, like other catalysts, act by lowering the free energy of activation for a reaction. A plot of the velocity of an enzyme-catalyzed reaction v versus the concentration of the substrate S is called a substrate saturation curve. The turnover number of an enzyme, kcat, is a measure of its maximal catalytic activity (the number of substrate molecules converted into product per enzyme molecule per unit time when the enzyme is saturated with substrate). Inhibition studies on enzymes have contributed significantly to our understanding of enzymes. Reversible inhibitors bind to the enzyme through noncovalent association/ dissociation reactions. Reversible inhibitors may bind at the active site of the enzyme (competitive inhibition) or at some other site on the enzyme (noncompetitive inhibition). Usually, enzymes catalyze reactions in which two (or even more) substrates take part, so the reaction is bimolecular. In singledisplacement reactions, both substrates, A and B, are bound before reaction occurs. In double-displacement (or pingpong) reactions, one substrate (A) is bound and reaction occurs to yield product P and a modified enzyme form, E9. The second substrate (B) then binds to E9 and reaction occurs to yield product Q and E, the unmodified form of enzyme. Early enzyme specificity studies by Emil Fischer led to the hypothesis that an enzyme resembles a "lock" and its particular substrate the "key.
|