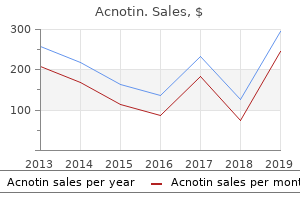
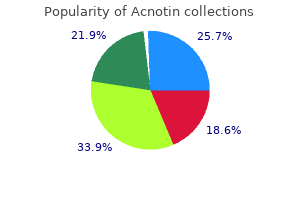
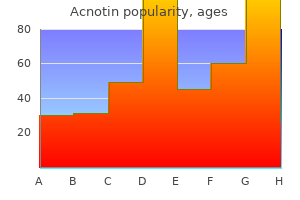
Acnotin"Purchase cheap acnotin on-line, skin care therapist". By: D. Lukjan, M.A., M.D., Ph.D. Medical Instructor, University of Connecticut School of Medicine Ambient measurements tend to be highest at roadside sites; some studies acne 39 weeks pregnant buy acnotin from india, but not all, report higher concentrations in vehicles than at roadside sites. Seasonally, the highest formaldehyde concentrations are associated with the highest rate of photochemical activity, and it appears that photochemical activity in summer contributes more formaldehyde to ambient concentrations than do direct vehicle emissions. While mobile sources are clearly important contributors to ambient concentrations of formaldehyde, indoor sources are the predominant source of exposure. Indoor concentrations are generally three to five times higher than outdoor concentrations. Indoor concentrations and personal exposures show seasonal trends, with higher concentrations in summer than winter. However, the role of seasonal variability in ambient concentrations in determining these seasonal trends in indoor concentrations is not clear. These classifications are based on both human and animal evidence that indicates a risk of nasopharyngeal cancer. Various risk assessments have been carried out for the purpose of defining acceptable exposure concentrations in occupational settings and in ambient air. Direct contact with tissues, such as those of the upper respiratory tract, can cause local irritation and acute and chronic toxic and genotoxic effects. In rats, after long-term inhalation, formaldehyde causes tumors in the nasal mucosa. After long-term oral administration, it causes hyperplasia and keratinization in the forestomach as well as inflammation and ulcers in the glandular stomach. Formaldehyde, at concentrations found in occupational settings, might be associated with myeloid leukemia, although the evidence for this is not sufficient to conclude that a causal relationship exists. There is limited evidence that exposure to formaldehyde in indoor air increases the occurrence of asthma symptoms in children. In workers with long-term exposure to formaldehyde, lesions in the nasal mucosa were observed at concentrations lower than 1. At present, there is little evidence that exposure to formaldehyde concentrations found in ambient air is hazardous. Indoor concentrations are higher than corresponding ambient concentrations and approximately the same as urban roadside and urban in-vehicle concentrations. In Brazil, studies have documented an increase in formaldehyde concentrations associated with the use of ethanol-based fuels and compressed natural gas. Ambient concentrations in Brazil have increased to the same range as the highest indoor concentrations recently measured in many countries. Formaldehyde causes irritation of the eyes and respiratory system, with substantial variation in individual responses. Formaldehyde has been classified as a human carcinogen by regulatory agencies, but the human evidence is weak and inconsistent. Ambient concentrations of formaldehyde are generally lower than those that cause irritation of the eyes and respiratory system. However, concentrations in certain outdoor environments, such as near roadways, can approach those at which sensitive people experience irritation. There is no evidence that ambient concentrations of formaldehyde cause any form of cancer. Whether these increased emissions will increase the risk of adverse effects on human health, including cancer, is unknown. Develop a monitoring network capable of tracking long-term aldehyde concentrations in ambient air because such an increase in the use of alcohols in fuel is likely. The use of noncancer endpoints as a basis for establishing a reference concentration for formaldehyde. Re: Mortality from lymphohematopoietic malignancies among workers in formaldehyde industries. Formaldehyde: Hazard Characterization and Dose-Response Assessment for Carcinogenicity by the Route of Inhalation. Temporal variation of carbonyl compound concentrations at a semi-rural site in Denmark. This would help in understanding the mechanism of tumor induction and in establishing biomarkers of formaldehyde exposure and effect.
To identify or validate genetic markers that may predict the natural history of disease acne with mirena buy 20 mg acnotin with amex, response to therapy, and/or tolerability of medical therapy through genetic discovery research. A Wilcoxon rank sum test will be used to explore differences between treatment groups in change in status from baseline to each of the timepoints. Results (p-values) will be presented, but should be interpreted with caution as multiple endpoints are being tested, and the study has not been powered to test these exploratory endpoints. For imputation of missing data in the quality of life data, please refer to Section 3. This more inclusive definition allows subjects without a genotype, as determined per protocol by the central laboratory, to be included in the efficacy analysis. All safety data (except for laboratory tests with results that were cancelled by the lab) will be included in data listings based on the safety analysis set. Adverse Event Severity Adverse events are graded by the investigator according to the Gilead Grading Scale for Severity of Adverse Events and Laboratory Abnormalities as specified in the clinical study protocol. The severity grade of events for which the investigator did not record severity will be categorized as "missing" for tabular summaries and data listings, and will be considered the least severe for the purposes of sorting for data presentation. Events for which the investigator did not record the relationship to study drug will be considered to be related to study drug for summary purposes. The event is treatment emergent if the month and year of onset (or year of onset) of the event meets both of the following criteria: the same as or after the month and year (or year) of the first dose of any study drug the same as or before the month and year (or year) of the 30th day after the last dose of any study drug Summaries of Adverse Events and Deaths 7. Laboratory results cancelled by the central laboratory will not be included in analysis. The Week 4 safety follow-up visit will be presented as an additional separate visit. Graded Laboratory Values the Gilead Grading Scale for Severity of Adverse Events and Laboratory Abnormalities will be used for assignment of toxicity grades to laboratory results for purposes of analysis as Grade 0, Grade 1 (mild), Grade 2 (moderate), Grade 3 (severe) or Grade 4 (potentially life threatening). Grade 0 includes all values that do not meet criteria for an abnormality of at least Grade 1. Some laboratory tests have laboratory toxicity criteria for both increased and decreased levels; analyses for each direction (ie, increased, decreased) will be presented separately. If the relevant baseline laboratory data are missing, then any abnormality of at least Grade 1 will be considered treatment emergent. This listing will include the complete laboratory test profile for each laboratory test with the graded result throughout the study. Values falling outside of the relevant reference range and/or meeting Gilead Grading Scale will be flagged, as appropriate, in the data listings. For individual laboratory tests, subjects will be counted once based on the most severe postbaseline values when the criterion is met. The denominator will be the number of subjects in the safety analysis set with at least one nonmissing postbaseline value for the test. In the case of multiple values in an analysis window, data will be selected for analysis as described in Section 3. The summary will be sorted alphabetically by drug class and then by decreasing total frequency within a class. For purposes of programming, any medication with a stop date that is on/prior to first dosing date or start date that is after the last dose of any study drug will be excluded from this summary. If a partial stop date is entered, then the month and year (if day is missing) or year (if day and month are missing) that is prior to the study drug start date will be excluded from the summary. If a partial start date is entered, then the month and year (if day is missing) or year (if day and month are missing) that is after the study drug stop date will be excluded from the summary. A listing of all concomitant medications reported during the study will be provided. The number and percent of subjects in each cross-classification group will be presented (subjects with a missing value at baseline or on- treatment will not be included in the denominator for percent calculation). Other Safety Measures A data listing will be provided for subjects who become pregnant during the study. Data from this study will be combined with data from other studies in a meta-population analysis using mixed-effect modeling techniques. Each score is calculated only if at least half of corresponding items are not missing.
Even asymptomatic patients demonstrate abnormal cardiorespiratory response to exercise which is best unmasked by Dobutamine stress test acne under a microscope purchase 30mg acnotin with amex. As we have discussed before in the hemodynamics section, the role of inotropes, vasodilators and beta-blockers is limited. A large left-to-right shunt produces volume overload and stress on the single ventricle. The rule of thumb is that if angiography of a systemic vessel gives rise to pulmonary capillary blush and opacification of pulmonary veins, it should be occluded percutaneously. One residual pulmonary arteriovenous malformation after transcatheter coil occlusion. Courtesy: Reprinted from reference 51 8 cyanotic Heart diSeaSeS lymphatic dysfunction60,61 Pathogenesis A Fontan circulation operates at/just beyond the functional limits of the lymphatic system. The superior vena caval pressure is elevated and its runoff decreased, impeding drainage of the thoracic duct. Leakage in the interstitium causes lymphedema or pulmonary edema, a very lethal complication in the early postoperative period. Leakage into the thorax or pericardium will lead to chylothorax or chylopericardium, a complication which usually only occurs in the perioperative period, but rarely thereafter because of adhesions. Leakage of chyle into the bronchus leads to plastic bronchitis, most frequently diagnosed at necropsy. The prognosis is very poor with 5 and 10 year survival rates of 59 and 20 percent, respectively. Increased systemic venous pressure, low velocity flow within the systemic venous atrium and pulmonary circulation, low cardiac output and dehydration contribute to the risk of thrombus formation. Massive pulmonary thromboembolism is the most common cause of sudden out-of-hospital death in patients with a Fontan circuit. Chronic multiple pulmonary microemboli may lead to pulmonary vascular obstructive disease, which may appear late but is particularly lethal in a Fontan circulation. There is no consensus, however, regarding the postoperative mode and duration of prophylactic anticoagulation, since no large scale randomized control studies have been performed. Routine anticoagulation with coumadin is performed by some institutions irrespective of the type of the modified Fontan procedure and potential risk factors. Keeping in mind the bimodal presentation of thromboembolism, our protocol is to administer oral anticoagulants for 1 year postsurgery, then switch over to oral antiplatelets and finally restart oral anticoagulants from 10 years post surgery onwards. An autoimmune or inflammatory cause, sometimes triggered by an infection, has also been isolated in some cases. A diet high in calories, high protein content and medium chain triglyceride fat supplements with low salt content is usually recommended. In some patients, specific anti-infection measures are necessary (chronic antibiotics, vaccines). Surgical correction of stenosed anastomotic sites, leaking atrioventricular valves, late takedown, etc. Cardiac transplantation with consequent immunosuppressive therapy has been tried in refractory cases. As explained before, treatment with inotropes, vasodilators, and diuretics show little result. Persistent segmental atelectasis, large airway obstruction or expectoration of tenacious mucoid material should prompt early diagnostic and therapeutic bronchoscopic lavage. However, normal pregnancy is associated with 30 to 40 percent increase in cardiac output and circulating blood volume and decrease in systemic vascular resistance by 24 weeks of gestation. These changes lead to increase in systemic venous pressure and may trigger right heart failure in a post-Fontan lady. The risk of right-to-left shunt, venous thrombosis and pulmonary embolism is increased. Studies reveal that an oxygen saturation of lesser than 85 percent was predictive of increased risk. The risk of the fetus having congenital heart disease is currently unknown, as women with cardiac malformations amenable to Fontan surgery have rarely had offspring. Nearly 10 percent of congenital cardiac malformations belong to functionally univentricular heart. The current therapy is a staged surgical approach called "Fontan palliation" which routes the systemic venous circulation to pulmonary circulation without an interposing ventricle.
Spread Squamous cell carcinoma has various grades of its differentiation or variants such as transitional cell carcinoma and lymphoepithelioma acne 4 week old baby purchase acnotin toronto. It may spread into the cranium through foramen lacerum and involve cranial nerves. Regional features: They include following: Nose: Unilateral nasal obstruction and blood stained discharge, denasal speech (rhinolalia clausa). Nodal metastases during first consultation are seen in 75% and about half of them have bilateral nodes. Type 1 Type 2 type 3 Undifferentiated carcinoma -Without lymphoid stroma -With lymphoid strom fig. Fine-needle aspiration cytology: Fine-needle aspiration cytology of cervical node may be required in some cases. Induction, adjuvant, concomitant, or their combinations have shown improvement in survival. Radical neck dissection may be done for persistent nodes when primary has been controlled. Pertinent anatomy: the orbit is related to frontal sinus (superiorly), maxillary sinus (inferiorly), ethmoid sinus (medially) and sphenoid sinus (posteromedially). Tumors of retro-orbital region Management: Clinical features, imaging studies and biopsy will confirm the diagnosis. Types: Dermoid with skin appendages (hairy polyp); True teratomas (have elements of all the three germ layers); and the epignathi (have well developed fetal parts). Nasopharyngeal angiofibroma: Most common site of origin is sphenopalatine foramen. This benign tumor erodes bone and is known for its recurrence on incomplete removal. If the mass is arising from the lateral wall of nose extending only into the nasopharynx, transpalatal approach is used. Other modalities of treatment include hormones and intensity modulated radiotherapy. Rhabdomyosarcoma: It is the most common malignant tumor of nasopharynx in children. Management of Juvenile Nasopharyngeal Angiofibroma: A Five Year Retrospective Study. Nasopharyngeal angiofibroma with cavernous sinus involvement-an unusual presentation. Neglected foreign body infratemporal fossa, a typical presentation: A case report. Any probable aetio-pathological factors that you know about for it to be juvenile We must learn that nothing can happen to us, unless make ourselves susceptible to it. The high and increasing incidence of oropharyngeal cancer is becoming a serious health problem. For the purpose of description of malignant tumors, oropharynx has been divided into four different subsites: 1. Exophytic: Superficially spreading and exophytic types are usually seen in the palatine arch. Ulcerative: Ulcerative and infiltrative types deeply invade the adjoining structures. Prognosis: Because of marked tendency for regional metastasis, prognosis is poor in ulcerative lesions than exophytic type of growth. It shows soft tissue infiltration such as tongue base, parapharyngeal and pre-epiglottic and bone marrow involvement. T3 and T4 lesions: Surgical excision with mandibular resection, neck dissection and postoperative radiation. T 4 lesion extending into anterior two-thirds-of tongue or vallecula: Extensive surgery consists of total glossectomy and laryngectomy in addition to the block dissection. Lymphatic: Fifty percent cases show cervical node (usually jugulodigastric) involvement at the time of initial presentation.
Approximately 1 to 4% of patients per year with established cirrhosis will progress to hepatocellular carcinoma 20195 skin care zarraz paramedical discount 10 mg acnotin visa, 20196, 20197, 20198. Complications of chronic hepatitis C account for the majority of liver transplantations in the United States 8114. The study met its primary endpoint of superiority over an historical control rate of 25% (p < 0. An interim analysis for safety and antiviral activity was conducted at 12 weeks on-treatment. Upon completion of an interim safety review of these subjects at Week 12 of treatment, Phase 2 was initiated in 50 subjects with any stage of fibrosis. Upon admission to the hospital, the subject was administered broad spectrum antibiotics. Fourteen subjects comprised of male and non-pregnant, non-lactating female subjects aged between 18 to 45 years old were enrolled and received study drug. One Grade 3 laboratory abnormality, 3+ blood in the urine, was reported in a 35 year old female. Furthermore, a relatively linear exposure-response relationship in the 20 to 80% maximal effect range is observed. During the conduct of the study the Sponsor will perform ongoing safety data review. All subjects will complete a 4-week and 12-week Post-Treatment visit regardless of treatment duration. The assessments performed at each visit are described in Section 6 and shown in Appendix 2. Final Amendment 1 Confirmation should be performed as soon as possible and must occur no later than 2 weeks after an initial observation indicating virologic failure during the on-treatment phase. Additionally, all subjects must complete the 4-week and 12-week Post-Treatment visits, based on their last dose date. Subsequent visits will be determined by the virologic response to treatment as outlined in Section 3. Inclusion Criteria Subjects must meet all of the following inclusion criteria to be eligible for participation in this study. Final Amendment 1 ii) Cirrhosis as defined as any one of the following: (1) Liver biopsy showing cirrhosis. Women 50 years of age with amenorrhea will be considered to be of childbearing potential. These women must have a negative serum pregnancy test at Screening and a negative urine pregnancy test on the Baseline/Day 1 visit prior to randomization. Exclusion Criteria Subjects who meet any of the following exclusion criteria are not to be enrolled in this study. Subjects with psychiatric illness (without the prior mentioned conditions) that is well-controlled on a stable treatment regimen for at least 12 months prior to randomization or has not required medication in the last 12 months may be included. Final Amendment 1 3) Pregnant or nursing female or male with pregnant female partner. Each bottle contains 30 tablets and a silica gel desiccant canister or sachet and polyester packing material. All drug products should be stored in a securely locked area, accessible only to authorized site personnel. Each subject must be given instructions to maintain approximately the same daily dosing interval between study drug doses. Subjects who are to receive total daily doses of 1000 mg, should be instructed to take 3 tablets in the morning and 2 tablets in the evening (or 2 tablets in the morning and 3 in the evening). Monitor for signs and symptoms of muscle weakness or myopathy, including rhabdomyolysis. All subjects will complete a 4-week and 12-week Post Treatment visit regardless of the treatment duration. Purchase acnotin online. Samhealthyskin skincare for men and women who looks and feels the best.
|